Welcome to OstomyCure AS
INTRODUCING THE TIES® SOLUTION
An innovative solution for ileostomy patients currently under clinical investigation.
- The aim of the TIES solution is that the patient can go about their normal daily life without the need to constantly wear an ostomy bag
- Developed and manufactured in accordance with regulatory standards to ensure the safety of patients
- Our aim is that patient should no longer feel discomfort. Over time, the implant is integrated into the soft tissue of the abdomen and becomes just like any other efficient, working part of your body
Life for many stoma patients is difficult at times when the stoma controls their lives. The new TIES Solution intends to not only make life easier but also to increase quality of life by helping those with a stoma to take back control and to live a more carefree life.
The TIES clinical trial is now accepting patients in the UK, Sweden, Austria, Poland and India.
Hospitals which are active in the clinical trial are located in London, Leeds, Glasgow, Oxford, Vienna, Brzezni, Warzawa, Linkoping, Gothenburg, Nagpur, Jaipur and Chandigarh. More clinics are in the pipeline and will be posted on our website as they commit, and our enrolment process is finalised. Contact us for updates.

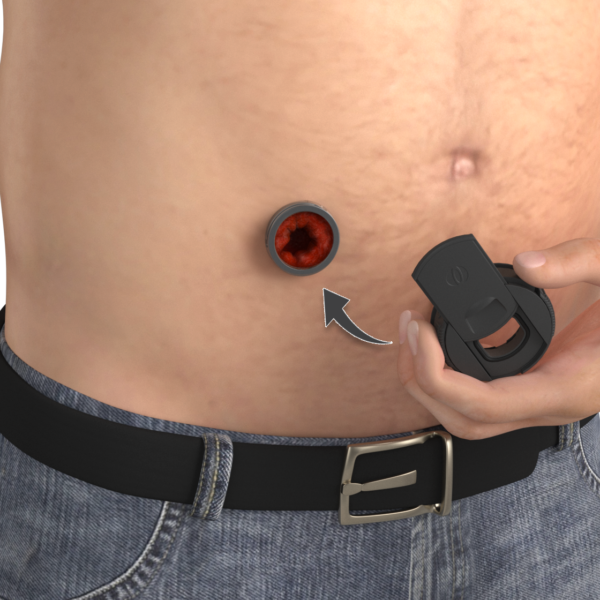
TIES® IMPLANT PORT
Integrates with the soft tissue of the abdomen, with optimized surfaces to promote healing.

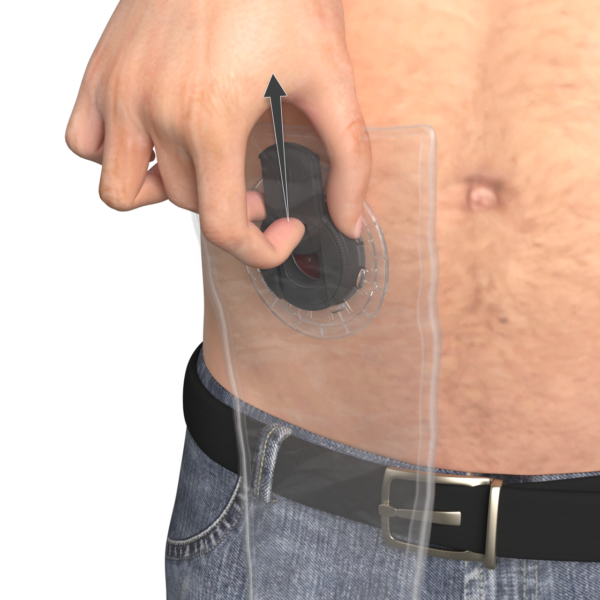
TIES® LID
No permanent bag, allowing a greater level of freedom and control.

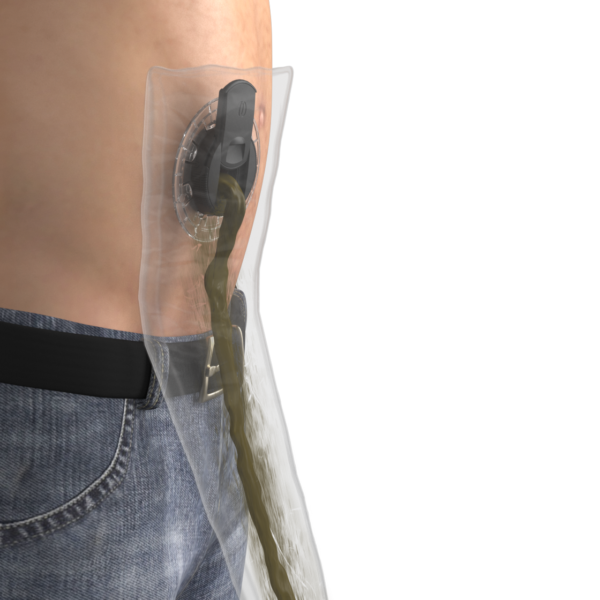
TIES® EMPTYING POUCH
A dedicated accessory to the Lid, used when emptying the intestinal waste.
TIES SYSTEM BENEFITS
TIES is the result of several years of research combined with the latest manufacturing technology.
The system is developed in Norway and Sweden, and is manufactured in accordance with current regulations to ensure patient safety.
-
Improved health economics
It is estimated that stoma patients spend around EUR 3000 a year on stoma bags and other consumables.
-
No leakage, odours or accidents
The TIES Lid seals the TIES Port, with a hatch that you can open and close to empty the stoma.
-
No permanent ostomy bag
No permanent bag means reduced risk of skin irritation, infections and other inconveniences.
-
Improved QoL
Aim to give patients their quality of life back using the latest technology, with no compromises on quality or safety.
-
Lower risk of hernia
Early indications that the TIES Solution lowers the risk of parastomal herniation
-
A simpler life
The ground breaking TIES Ileostomy system helps stoma patients take back control and live a more carefree life.
CLINICAL PARTNERS
The TIES clinical trial is now accepting patients in the UK, Sweden, Austria, Poland and India.
- CONTACT US FOR DETAILS
JOIN OUR CLINICAL STUDY
Are you and your clinic interested in being part of the TIES study?
Please get in contact with us for further details- REGISTER FOR STUDY